Sidebar
 This version (2019/02/13 17:46) is a draft.
This version (2019/02/13 17:46) is a draft.Approvals: 0/1
This is an old revision of the document!
Table of Contents
Overview
CrYOLO is a fast and accurate particle picking procedure. It's based on convolutional neural networks and utilizes the popular You Only Look Once (YOLO) object detection system.
- crYOLO makes picking fast – On a modern GPU it will pick your particles at up to 6 micrographs per second.
- crYOLO makes picking smart – The network learns the context of particles (e.g. not to pick particles on carbon or within ice contamination )
- crYOLO makes training easy – You might use a general network model and skip training completely. However, if the general model doesn't give you satisfactory results or if you would like to improve them, you might want to train a specialized model specific for your data set by selecting particles (no selection of negative examples necessary) on a small number of micrographs.
In this tutorial we explain our recommended configurations for single particle and filament projects. You can find more information about supported networks and about the config file in the following articles:
Installation
You can find the download and installation instructions here: Download and Installation
Picking - Using a model trained for your data
Data preparation
CrYOLO supports MRC, TIF and JPG files. It can work with 32 bit data, 8 bit data and 16 bit data. It will work on original MRC files, but it will probably improve when the data are filtered. Therefore you should low-pass filter them to a reasonable level. Since Version 1.2 crYOLO can automatically do that for you. You just have to add
"filter": [0.1,"filtered"]
to the model section in your config file to filter your images down to an absolute frequency of 0.1. The filtered images are saved in folder filtered.
If you followed the installation instructions, you now have to activate the cryolo virtual environment with
source activate cryolo
In the following I will assume that your image data is in the folder full_data.
The next step is to create training data. To do so, we have to pick single particles manually in several micrographs. Ideally, the micrographs are picked to completion. One may ask how many micrographs have to be picked? It depends! Typically 10 micrographs are a good start. However, that number may increase / decrease due to several factors:
- A very heterogenous background could make it necessary to pick more micrographs.
- If your micrograph is only sparsely decorated, you may need to pick more micrographs.
We recommend that you start with 10 micrographs, then autopick your data, check the results and finally decide whether to add more micrographs to your training set.
 To create your training data, crYOLO is shipped with a tool called “boxmanager”. However, you can also use tools like e2boxer to create your training data.
To create your training data, crYOLO is shipped with a tool called “boxmanager”. However, you can also use tools like e2boxer to create your training data.
Start the box manager with the following command:
cryolo_boxmanager.py
Now press File → Open image folder and the select the full_data directory. The first image should pop up. You can navigate in the directory tree through the images. Here is how to pick particles:
- LEFT MOUSE BUTTON: Place a box
- HOLD LEFT MOUSE BUTTON: Move a box
- CONTROL + LEFT MOUSE BUTTON: Remove a box
You can change the box size in the main window, by changing the number in the text field labeled Box size:. Press Set to apply it to all picked particles. For picking, you should the use minimum sized square which encloses your particle.
If you finished picking from your micrographs, you can export your box files with Files → Write box files.
Create a new directory called train_annotation and save it there. Close boxmanager.
Now create a third folder with the name train_image. Now for each box file, copy the corresponding image from full_data into train_image. crYOLO will detect image / box file pairs by search taking the box file an searching for an image filename which contains the box filename.
Configuration
You now have to create a config file your picking project. To do this type:
touch config.json
To use the Phosaurus network copy the following lines into that file:
- config.json
{ "model" : { "architecture": "PhosaurusNet", "input_size": 1024, "anchors": [160,160], "max_box_per_image": 600, "num_patches": 1, "filter": [0.1,"filtered"] }, "train": { "train_image_folder": "train_image/", "train_annot_folder": "train_annotation/", "train_times": 10, "pretrained_weights": "model.h5", "batch_size": 6, "learning_rate": 1e-4, "nb_epoch": 50, "warmup_epochs": 0, "object_scale": 5.0 , "no_object_scale": 1.0, "coord_scale": 1.0, "class_scale": 1.0, "log_path": "logs/", "saved_weights_name": "model.h5", "debug": true }, "valid": { "valid_image_folder": "", "valid_annot_folder": "", "valid_times": 1 } }
Click here to get more information about the configuration file
Please set the value in the “anchors” field to your desired box size. It should be size of the minimum enclosing square in pixels. Furthermore check if the fields “train_image_folder” and “train_annot_folder” have the correct values. Typically, 20% of the training data are randomly chosen as validation data. If you want to use specific images as validation data, you can move the images and the corresponding box files to the folders specified in “valid_image_folder” and “valid_annot_folder”. Make sure that they are removed from the original training folder! With the line below, crYOLO automatically filters your images to an absolute frequence 0.1 and write them into a folder “filtered”.
"filter": [0.1,"filtered"].
crYOLO will automatically check if an image in full_data is available in the filtered directory. The filtering is done in parallel. If you don't want to use crYOLO's internal filtering, just remove the line and filter them manually. If you remove the line, don't forget to remove the comma at the end of the line above.
Please note the wiki entry about the crYOLO configuration file if you want to know more details.
Training
Now you are ready to train the model. In case you have multiple GPUs, you should first select a free GPU. The following command will show the status of all GPUs:
nvidia-smi
For this tutorial, we assume that you have either a single GPU or want to use GPU 0. Therefore we add '-g 0' after each command below. However, if you have multiple (e.g GPU 0 and GPU 1) you could also use both by adding '-g 0 1' after each command.
Navigate to the folder with config.json file, train_image folder, etc.
1. Warm up your network
cryolo_train.py -c config.json -w 3 -g 0
2. Train your network
cryolo_train.py -c config.json -w 0 -g 0
The final model will be called model.h5
The training stops when the “loss” metric on the validation data does not improve 5 times in a row. This is typically enough. However, you might want to give the training more time to find the best model. You might increase the “not changed in a row” parameter to, for example, 10 by adding the flag -e 10:
cryolo_train.py -c config.json -w 0 -g 0 -e 10
to the training command.
Picking
You can now use the model weights saved in model.h5 to pick all your images in the directory full_data. To do this, run:
cryolo_predict.py -c config.json -w model.h5 -i full_data/ -g 0 -o boxfiles/
You will find the picked particles in the directory boxfiles
If you want to pick less conservatively or more conservatively you might want to change the selection threshold from the default of 0.3 to a less conservative value like 0.2 or more conservative value like 0.4 using the -t parameter:
cryolo_predict.py -c config.json -w model.h5 -i full_data/ -g 0 -o boxfiles/ -t 0.2
Visualize the results
To visualize your results you can use the box manager:
cryolo_boxmanager.py
Now press File → Open image folder and the select the full_data directory. The first image should pop up. Then you import the box files with File → Import box files and select in the boxfiles folder the EMAN directory.
Picking - Without training using a general model
The general model can be found here: Download and Installation.
Configuration
The next step is to create a configuration file. Type:
touch config.json
Open the file with your preferred editor.
For the general Phosaurus network enter the following inside:
- config.json
{ "model" : { "architecture": "PhosaurusNet", "input_size": 1024, "anchors": [205,205], "max_box_per_image": 700, "num_patches": 1, "filter": [0.1,"tmp_filtered"] } }
Please set the value in the “anchors” field to your desired box size. It should be size of the minimum particle enclosing square in pixel.
Picking
Just follow the description given above
As for a direct trained model, you might want to play around with the -t parameter to make picking less or more conservative.
Picking filaments - Using a model trained for your data
Since version 1.1.0 crYOLO supports picking filaments.
Filament mode on Actin:
Filament mode on MAVS (EMPIAR-10031) :
Data preparation
 As described previously, filtering your image using a low-pass filter is probably a good idea.
As described previously, filtering your image using a low-pass filter is probably a good idea.
After this is done, you have to prepare training data for your model. Right now, you have to use the sxhelixboxer.py to generate the training data:
sxhelixboxer.py --gui my_images/*.mrc
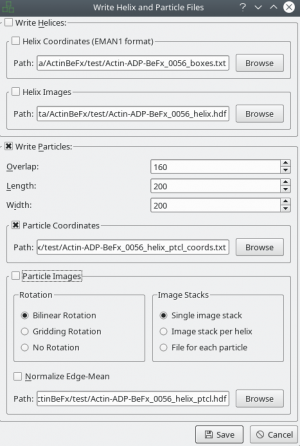
After tracing your training data in sxhelixboxer, export them using File → Save. Make sure that you export particle coordinates as this the only format supported right now (see screenshot). In the following example, it is expected that you exported into a folder called “train_annotation”.
Configuration
You can configure it the same way as for a “normal” project. We recommend to use YOLO in "patch mode" with 3x3 patches:
- config.json
{ "model" : { "architecture": "YOLO", "input_size": 768, "anchors": [200,200], "max_box_per_image": 600, "num_patches": 3, "filter": [0.1,"tmp_filtered"] }, "train": { "train_image_folder": "train_image/", "train_annot_folder": "train_annotation/", "train_times": 10, "pretrained_weights": "model.h5", "batch_size": 6, "learning_rate": 1e-4, "nb_epoch": 50, "warmup_epochs": 0, "object_scale": 5.0 , "no_object_scale": 1.0, "coord_scale": 1.0, "class_scale": 1.0, "log_path": "logs/", "saved_weights_name": "model.h5", "debug": true }, "valid": { "valid_image_folder": "", "valid_annot_folder": "", "valid_times": 1 } }
Click here to get more information about the configuration file
Just adapt the anchors accordingly to your box size.
Training
In principle, there is not much difference in training crYOLO for filament picking and particle picking. For project with roughly 20 filaments per image we successfully trained on 40 images (⇒ 800 filaments). However, in our experience the warm-up phase and training need a little bit more time:
1. Warm up your network
cryolo_train.py -c config.json -w 10 -g 0
2. Train your network
cryolo_train.py -c config.json -w 0 -g 0 -e 10
The final model will be called model.h5
Picking
The biggest difference in picking filaments with crYOLO is during prediction. However, there are just three additional parameters needed:
- - -filament: Option that tells crYOLO that you want to predict filaments
- -fw: Filament width (pixels)
- -bd: Inter-Box distance (pixels).
Let's assume you want to pick a filament with a width of 100 pixels (-fw 100). The box size is 200×200 and you want a 90% overlap (-bd 20). Moreover, you wish that each filament has at least 6 boxes (-mn 6). The micrographs are in the full_data directory. Than the picking command would be:
cryolo_predict.py -c config.json -w model.h5 -i full_data --filament -fw 100 -bd 20 -o boxes/ -g 0 -mn 6
The directory boxes will be created and all results are saved there. The format is the eman2 helix format with particle coordinates. You can find a detailed description how to import crYOLO filament coordinates into Relion here.
Visualize the results
You can use the boxmanager as described previously.
Evaluate your results
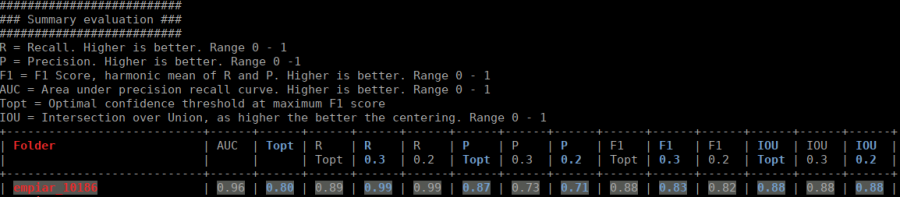
The evaluation tool allows you, based on your validation data, to get statistics about your training. Unfortunately, this script does not work for filamental data. If you followed the tutorial, the validation data are selected randomly. With crYOLO 1.1.0 a run file for each training is created and saved into the folder runfiles/ in your project directory. This run file contains which files were selected for validation, and you can run your evaluation as follows:
cryolo_evaluation.py -c config.json -w model.h5 -r runfiles/run_YearMonthDay-HourMinuteSecond.json -g 0
The table contains several statistics:
- AUC: Area under curve of the precision-recall curve. Overall summary statistics. Perfect classifier = 1, Worst classifier = 0
- Topt: Optimal confidence threshold with respect to the F1 score. It might not be ideal for your picking, as the F1 score weighs recall and precision equally. However in SPA, recall is often more important than the precision.
- R (Topt): Recall using the optimal confidence threshold.
- R (0.3): Recall using a confidence threshold of 0.3.
- R (0.2): Recall using a confidence threshold of 0.2.
- P (Topt): Precision using the optimal confidence threshold.
- P (0.3): Precision using a confidence threshold of 0.3.
- P (0.2): Precision using a confidence threshold of 0.2.
- F1 (Topt): Harmonic mean of precision and recall using the optimal confidence threshold.
- F1 (0.3): Harmonic mean of precision and recall using a confidence threshold of 0.3.
- F1 (0.2): Harmonic mean of precision and recall using a confidence threshold of 0.2.
- IOU (Topt): Intersection over union of the auto-picked particles and the corresponding ground-truth boxes. The higher, the better – evaluated with the optimal confidence threshold.
- IOU (0.3): Intersection over union of the auto-picked particles and the corresponding ground-truth boxes. The higher, the better – evaluated with a confidence threshold of 0.3.
- IOU (0.2): Intersection over union of the auto-picked particles and the corresponding ground-truth boxes. The higher, the better – evaluated with a confidence threshold of 0.2.
If the training data consists of multiple folders, then evaluation will be done for each folder separately. Furthermore, crYOLO estimates the optimal picking threshold regarding the F1 Score and F2 Score. Both are basically average values of the recall and prediction, whereas the F2 score puts more weights on the recall, which is in the cryo-em often more important.
Advanced parameters
During training (cryolo_train), there is the following advanced parameter:
- –warm_restarts: With this option the learning rate is decreasing after each epoch and then reset after a couple of epochs.
During picking (cryolo_predict), there are two advanced parameters:
- -t confidence_threshold: With the -t parameter, you can let the crYOLO pick more conservative (e.g by adding -t 0.4 to the picking command) or less conservative (e.g by adding -t 0.2 to the picking command). The valid parameter range is 0 to 1.
- -d distance_in_pixel: With the -d parameter you can filter your picked particles. Boxes with a distance (pixel) less than this value will be removed.
- -pbs prediction_batch_size: With the -pbs parameter you can set the number of images picked as batch. Default is 3.
Help
Any questions? Problems? Suggestions?
Find help at our mailing list!